What is Radioactive Decay Inside Earth?
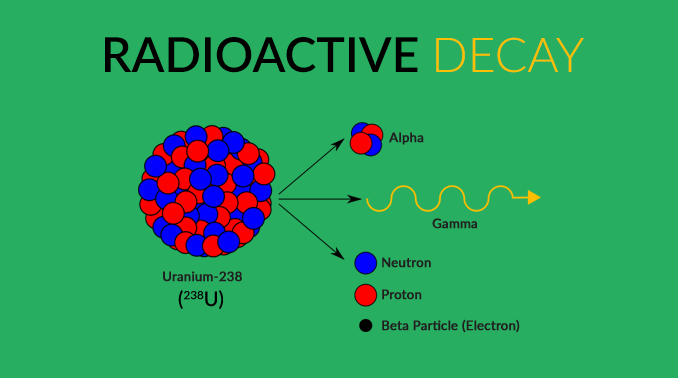
“Four radioactive isotopes inside Earth account for about 50% of Earth’s internal heat. Like a slow cooker, they constantly release heat within the planet keeping it on a light simmer.”
These four isotopes are:
- Uranium-238 (238U)
- Uranium-235 (235U)
- Thorium-232 (232Th)
- Potassium-40 (40K)
The majority of the heat transfer occurs at mid-oceanic ridges. Whereas the least amount of heat transfer is from the continental interiors.
Are radioactive isotopes a source of energy for plate tectonics?
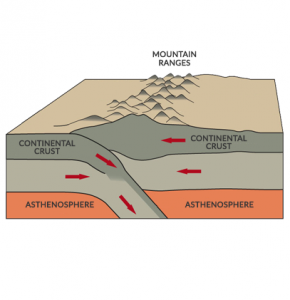
Certain isotopes of elements are unstable and radioactive. For example, uranium, thorium, and potassium isotopes are deep inside Earth.
These radioactive isotopes generate 50% of Earth’s radiogenic heat from radioactive decay. The remaining 50% of Earth’s internal heat budget is from primordial heat after its initial formation.
It’s this radioactive heat in the mantle that makes our planet geologically active. The majority of internal heat transfer occurs volcanically at mid-oceanic ridges. This process drives mantle convection and plate tectonic motion on the planet.
These radioactive isotopes have long lifetimes before they decay and release slow amounts of energy. It’s because of these 4 isotopes that Earth maintains a cozy temperature in the mantle.
How does radioactive decay heat the asthenosphere?
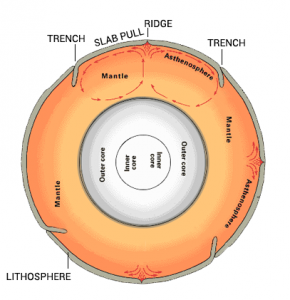
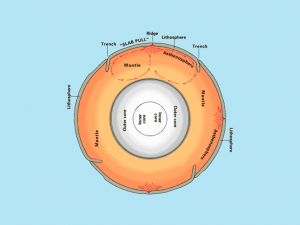
Now that we have a heat source inside Earth, it’s the asthenosphere where the convection cycle occurs because particles can freely flow.
But in our rigid lithosphere which is a solid, particles cannot freely move.
As heat rises, it starts a mantle convection cycle. It tears apart the Earth to form mid-oceanic ridges (tensional force). When it sinks down, it breaks it apart (compressional force).
SLAB PULL: Slab pull is the main mechanism pulling the lithosphere apart at divergent plate boundaries.
What are slab pull and plate tectonics?
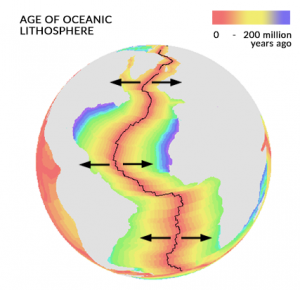
Because of the convection cycles that occur deep inside Earth, it’s the force that breaks apart the whole lithosphere into 7 major plate tectonics and 12 or so minor ones.
- DIVERGENT PLATES: At divergent plate boundaries, plates pull apart from each other. In turn, lava spews out to create the youngest geological rocks on Earth.
- CONVERGENT PLATES: At the opposite end, convergent plate boundaries smash into each other like a demolition derby. The force is so incredible that it causes volcanoes, earthquakes, and mountain building events.
It’s this radioactive heat that powers the rock cycle and plate tectonics on Earth.
How do nuclear power plants use radioactive decay?
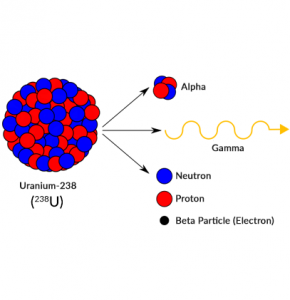
We use uranium and thorium in nuclear power plants in order to make fuel for fission reactors. Because of the possibility of overheating, nuclear power plants are usually located close to a source of water to cool down.
Certain elements have isotopes with a specific number of neutrons such as uranium-238 (238U). Each number corresponds to the number of protons and neutrons in its nucleus.
Overall, it’s the weak nuclear force that is responsible mostly for decay. For example, this force can change an electron into a neutrino, and a neutron into a proton. Through this force, atoms will decay or end up unstable and radioactive.
What is Radioactive Decay Inside Earth?
Earth is a radioactive planet. It contains traces of uranium, thorium, potassium, and other radioactive elements. These are the building blocks for radioactive decay inside Earth.
This continuous process of radioactive decay generates heat within the Earth’s core, contributing to the planet’s internal energy budget and influencing geological processes such as mantle convection and plate tectonics.
As always, if you have any comments or questions, make sure to leave them in the comment form below. We’ll do our best to get back to you and answer as soon as we can.








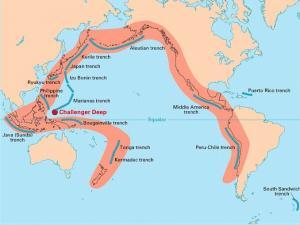

The Pacific major plate is the largest which underlies the Pacific Ocean. Specifically, it stretches all the way along the west coast of North America to the east coast of Japan and Indonesia.
Thanks for taking my question. What is the “endgame” of the radioactive materials inside the Earth? In other words, what will they ultimately become and how will they then interact with the rest of the universe?
I don’t have to worry about this cycle drastically changing someday, right