Earth’s Atmosphere Composition: Nitrogen, Oxygen, Argon and CO2
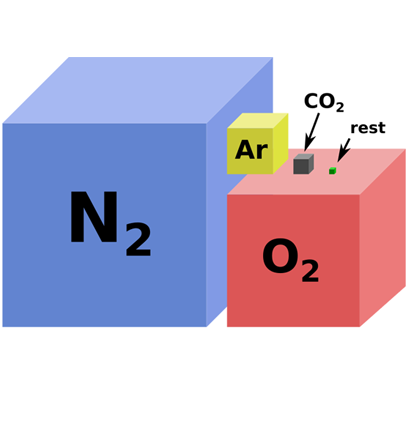
Some people are surprised to learn that oxygen isn’t the most abundant gas in Earth’s atmosphere composition.
Based on the relative volumes of the gases in Earth’s atmosphere, nitrogen is actually more than 3 times more than oxygen.
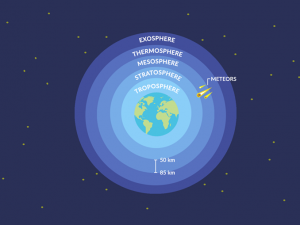
Because the troposphere is the lowest atmosphere layer, it contains 75 percent of the atmosphere’s mass.
From largest to smallest, Earth’s atmosphere composition contains nitrogen, oxygen, argon, CO2, and trace gases. Because water vapor is highly variable geographically, it’s excluded from this total.
1. Nitrogen (78.1%)

While nitrogen is the most abundant gas in Earth’s atmosphere, it only makes up 0.005% of Earth’s crust in weight (David Darling).
Nitrogen is incredibly stable and requires a lot of energy to change forms.
Even though its volume in Earth’s crust is relatively small, nitrogen plays an important role in the nitrogen cycle.
As part of this cycle, nitrogen constantly exchanges between the atmosphere and living organisms.
2. Oxygen (20.9%)
Earth has the conditions for life to flourish. Oxygen is essential to human life as our lungs respire oxygen and uses it in metabolism.
While nitrogen is an extremely stable gas, it’s difficult to break up and use for chemical processes. But oxygen will readily take part in chemical reactions because it’s an electron thief.
So even though nitrogen is plentiful, we need oxygen to drive chemical reactions that produce energy.
3. Argon (0.93%)

As an inert gas, argon doesn’t bond or do much in the atmosphere.
This is why there’s no argon cycle. But we have nitrogen and carbon because of their ability to bond with other elements.
When potassium radioactively decays, argon is one of the possible products. And the lithosphere has lots of potassium.
It’s not too exciting of a gas. But it’s up in the atmosphere at 0.93% of air volume.
4. Carbon Dioxide (0.04%)
Carbon is the most important element for building molecules essential for living things.
As you can see from the long-term carbon cycle, carbon takes up various forms such as carbon dioxide (CO2), methane (CH4), and glucose (C6H12O6).
Since 1900, carbon dioxide has increased mostly because of human activity. After extracting fossil fuels, humans burn fossil fuels.
In turn, gases like methane and carbon dioxide become air pollution in the atmosphere. In fact, carbon dioxide has nearly doubled since 1900.
5. Trace Gases
The remaining portion of the atmosphere belongs to trace gases. For example, neon, helium, methane, methane, and krypton are some of the major trace gases that make up a small part of the atmosphere.
But humans can also cause some trace gases. For example, chlorofluorocarbons (CFCs) has damaged the ozone layer in the north and south pole.
When chlorine enters the troposphere and eventually the stratosphere, it reacts with ozone (O3) essentially depleting it. Similar to ozone, water vapor is a variable gas.
6. Water Vapor (Variable)
Water vapor has been removed from the 100% total because of its region variability. But it can make up large portions of the atmosphere. For example, it can make up 5% by volume in hot regions but much less in colder regions.
Water vapor regulates air temperature because it absorbs solar radiation. It evaporates from lakes and rivers on the surface of Earth. Once it’s in the atmosphere, water vapor condenses such as in the form of rain. It simply changes form from water vapor to a liquid.
As part of the hydrological cycle, water is always in motion. And it’s all driven by the sun’s energy.
The main focus is how water is stored – in the atmosphere, glaciers, oceans, plants, and humans. Most evaporation takes place in oceans. It’s the Coriolis effect that moves it away from the equator.
What is the distribution of gases in the atmosphere?
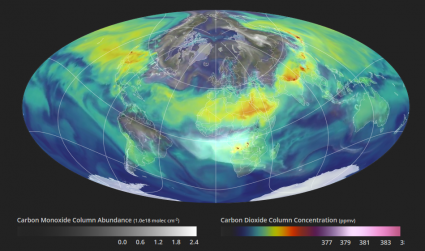
In this video, it displays a year in the life of Earth’s carbon dioxide. As you can see, carbon dioxide is the most important gas affected by human activity.
In the northern hemisphere, we see the highest concentrations of carbon dioxide from major emission sources. For example, carbon emissions are mostly focused around North America, Europe, and Asia. But the gas disperses, finding its circulation path with global weather patterns and ocean currents.
Even the seasonal patterns on Earth affect the amount of carbon dioxide in the atmosphere. During photosynthesis in spring and summer, plants absorb a substantial amount of carbon dioxide from the atmosphere.
As summer transitions to fall, photosynthesis begins to decrease as carbon dioxide accumulates back into the atmosphere. This effect is from the Earth’s metabolism or net primary productivity.
Earth’s Atmosphere Composition
The Earth’s atmosphere is made up of many different gases. At the lower levels, nitrogen and oxygen are the most abundant gases.
One of the most important things to remember about Earth’s atmosphere is that it is dynamic, meaning what we see today may not be the same as what we see years from now.
Do you have any comments or questions? Please let us know in the comment form below.







This is a hoax to get certain rich people make billions off of green energy, even if it effects the planet in a negative way. There are less particles per million. This allows more sunlight to make it to our surface. I’m older so I have seen and been effected by the sun. I get sun burned in 15 minutes compared to 40 years ago. The makers of sun block are racking in the money. Have you notice how high the spf has gone way higher now then when it came out. We are seeing alot more powerful hurricanes and record numbers of twisters. Used to get a lot of snow now we hardly get any. Snow is important to protect plants that are perennials
CO2 causes global warming. Global warming is very dangerous. Global warming will kill all of the worlds life. We will all burn up. All of this could be believable except for one fact.
Not to long ago 1/2 of the world was covered in ice over 1 mile thick. It was so cold nothing could grow or live here. Anyone with a little common sense should realize if it wasn’t for global warming the ice cover would not have melted.
Bruce W
Jan. 19 2025
Methane is listed twice. Hurray for the atmosphere, giver of oxygen & life.
water vapour is the largest greenhouse gas and has more effect in controlling worldwide temperatures. This is inconvenient to the green lobby and thus totally ignored.
With ‘TERROR’ of excessive CO2 what is today’s breakdown? Or is that information ‘CONTROLLED’?
Why is methane listed twice as a trace gas?
earthhow wrote:
“The greenhouse effect is what traps heat globally and causes more overall warming.”
Wrong. The “greenhouse effect” is a manifestation of the wholly-fictive “backradiation”, which is a manifestation of the misuse of the Stefan-Boltzmann equation in Energy Balance Climate Models (EBCMs), which I prove using the Kiehl-Trenberth ‘Earth Energy Balance’ graphic (which is a graphical representation of the results of the mathematics in their EBCM).
Climatologists use the idealized blackbody form of the S-B equation upon real-world graybody objects in their EBCMs. The idealized blackbody form of the equation assumes emission to 0 K and thus it inflates radiant exitance for all calculated-upon objects, which conjures “backradiation” out of thin air. This “backradiation” is claimed to cause the “greenhouse effect”, which is used to designate polyatomics as “greenhouse gases”, which is used to claim certain of those polyatomics cause CAGW (Catastrophic Anthropogenic Global Warming, due to CO2).
Except “backradiation” implies energy spontaneously flowing up an energy density gradient, in violation of 2LoT in the Clausius Statement sense. “Backradiation” violates the fundamental physical laws… and if a physical process violates the fundamental physical laws, it is physically impossible. All of the offshoots of CAGW (net zero, carbon credit trading, carbon capture and sequestration, etc.) are based upon a physical impossibility. We are literally chucking money into a fire with this nonsense.
IOW, CAGW is nothing more than a complex mathematical scam. I unwind that scam and irrefutably prove with mathematical precision that it is nothing more than a scam at the link below:
https://www.patriotaction.us/showthread.php?tid=2711
Since 1900, carbon dioxide has increased mostly because of human activity. After extracting fossil fuels, humans burn fossil fuels.
This Is a LIE That The Progressive Loony Left Wants You To Believe. When John D. Rockefeller got into the Oil Business, he wanted people to believe that Oil was scarce, it is in fact the second most available liquid in the world other than water, and there is a lot of it, it’s not scarce, and thus why there’s an oil cartel to make sure they don’t release too much at one time and cheapen it’s value.
CO2 Has NOT Increase Due to the Activity of Man, in the PAST there were higher levels of CO2, and the Animals and some plants tended to be much larger, and there were more varieties. But the Earth has Changed over the millenia, and MAN, has very little to do with the Changes in evolution when he isn’t busy detonating Nuclear Bombs, it’s a natural cycle.
Weather CAN be manipulated by Man with Geo Engineering (Chem Trails) and the Use of HAARP, which the DOD denies they still use, but they do, and they use it to assist Nature in the development of Hurricanes and Tornados, and can steer them somewhat with HAARP by shooting Billions of Watts of Microwave Radiation into our Ionosphere from one or more locations which bubbles the Atmosphere. You can see they are doing it sometime when you see clouds shaped in ripple/lines across the sky like waves at the beach.
There are Lies here.
For example…
Since 1900, carbon dioxide has increased mostly because of human activity. After extracting fossil fuels, humans burn fossil fuels.
Ok so no accounting for the fact that we did not really measure it in 1900? What samples and data are we basing this upon specifically? I think some more research is needed here. And 1900? That may as well be like yesterday in geological terms. There are natural events that can put out more Carbon in an afternoon then Humanity has in the last 100 years. And yes the planet warms and cools and always has for a number of reasons. So what they OMIT is as important as what they say here.
Secondly, calling hydrocarbons FOSSIL FUELS is an incorrect assertion. There is a moon of Saturn covered in Oceans of Methane. It has no life. Never had any Dinosaurs. Yet its covered in this stuff. So I think suggesting our Oil comes from dead Dinosaurs and Plants is incorrect. It may contribute, but we see Hydrocarbons produced other ways in our own Solar System.
It would not be a surprise if the Political Left was telling all kinds of lies about CARBON because it is an ideal tool for Billionaires to exert a much higher level of control and restrictions on humanity and human freedoms as well as serve as a scaffold for global taxation by a burgeoning would be totalitarian world government. Like all the social causes of the political left, it appears hijackecked via Oligarch controlled NGO’s and special interests. People with private jets and yachts moralizing at us for driving our little Toyotas to work. Its all BS. Just like every other thing the LEFT and ESTABLISHMENT says. BS. All of it. They want to keep us afraid, distracted, fighting with other and and demoralized while they steal the entire world.
Interesting… sounds all hell and brimstone… until you look at facts. The earth’s CO2 level is currently 0.04%, up from 0.03%. (not doubled). At 0.02% plant life dies on planet earth, so we know CO2 hasn’t doubled. Curious to see if EarthHow has the constitution to post a chart showing CO2 levels since the 1600’s (just CO2, not made up formula and only through current (facts not scare tactics). This chart changes the conversation. Yes, pollution is bad. Stop cutting down trees, all the reasonable things. Just don’t misrepresent data to try and scare us. We are smarter than that.
You explain that “Water vapor regulates air temperature because it absorbs solar radiation.” This is interesting, since water vapour responds to radiation across a broad range of frequencies, including incoming solar radiation. What more can you say about how exactly it regulates the global air temperature, and how its tropospheric effects differ in the stratosphere?
I’d be grateful to know more.
Thanks.
Curious that my comment was removed…
The graphic representation of the abundance of gasses at the top of the page, while accurate, can be misleading to anyone reading it as a 2D representation. A pie chart or other 2D graphic would be less misleading.
Yes, you’re correct. When we use energy, whether it’s from burning fossil fuels, generating electricity, or industrial processes… a large amount of that energy is transformed into heat. This is based on the idea of thermodynamics and the conservation of energy. These human activities is a big factor for generating heat at local levels…
But higher greenhouse gas concentrations has been the biggest contributor to higher global temperatures at a global scale. This includes everything from burning of fossil fuels for energy, deforestation, and other industrial activities. The greenhouse effect is what traps heat globally and causes more overall warming.
What if the increase in the global temperatures played hand in hand with the amount of energy the world uses each year. I’m not the rocket scientist but doesn’t energy give off heat when its either made or created?
Question: Have their been any studies on the geocorona vs the earth’s heating cooling issues?
Other than flat-out lies CO2 has doubled. (compare to (false) ice core data then this claim is a stretch as the ACTUAL ice core study has readings way over what there is today.
The hole in the Ozone layer was reported in the South (despite CFCs being abundant in the North.
That and the ice age that didn’t happen in the 70s or 30+ more prediction alarmist fails since..a great article?
No. Since there is no additional warming on any planet in our solar system with a thick atmosphere based on gas content and that CO2 increase is 95% due to temperature rise, not humans AND that CO2 was more than today just 12,000 years ago.
I needed this for an Earth Science project. It helped me so much. Thank you!!
I’m late to the party but I have to ask. How did scientists measure CO² in 1900? Is it possible that their instrumentation wasn’t quite advanced as today’s is and they could’ve been off by 1 or 2 100ths? And if CO2 blocks radiation from escaping the earth, wouldn’t it also block radiation from reaching the earth at the same rate? Or is there something about the molecules that only allow it to move one way and not the other?
My 11th edition of the Britannica places CO2 at .035% in 1911 but others back then speak of around .04%. Others mention that CO2 percentages vary all over the world, even by time of day and season but generally in the range now of .0415%, which is so small it could not have been measured a hundred years ago.
Here is a good visual; a 10 gallon kettle will hold 1,000 golf balls. If compared to the atmosphere 781 of those golf balls would be made of Nitrogen, 209 golf balls would represent Oxygen, 9 golf balls would equate to Argon and at best total CO2 would be represented by a tiny slice of paint chipped out from a single golf ball, the rest of which would be added to the 9 balls that make up Argon.
To the best of my knowledge “global warming” as we are told of it being aggrandized by increased CO2 in minute amounts has never been proven by scientific observation, and use of ancient bubbles of air in sea ice to establish a base for CO2 eons ago ignores the established fact that CO2 is quickly absorbed into water, and the colder the water is the faster the CO2 is absorbed.
Global Warming as taught today is a colossal HOAX.
You know what. Just try a way to include a reference
James Hansen said the CO2 feedback is only 0.007 W/m2 per decade, which is less energy than a bee requires to fly. The claim that CO2 drive climate is absurd.
Did you know that the remaining gases are each 0.005, it means that the product of these gases should be able to make a sun or maybe it is because it is made up of many other gases because of hydrogen plus helium = to the sun.
Interesting debate, but what immediately becomes clear as that the scapegoat of carbon is a precarious argument amongst real scientists. As one comment rightly said, even Patrick Moore, a founding member of Greenpeace, came to the realization that their scaremongering may not be founded on any real science or truth. The 0.04%, minute proportions of CO2, approximately 400 ppm is the only food that can be changed into foliage, hence the only food for plants and hence for humanity.
At 0.04% of the atmosphere will let you think for yourselves if a gas can have such an impact. Must be pretty toxic, yes i just exhaled some of it a sec ago! At 0.02% CO2 plants die! and industrial greenhouses pump it x5 if they want increased yield. Even Patrick Moore one of the greenpeace founders talks about the CO2 scam. There are many scientists on the logical side, check youtube.
Thank you. This is really helpful for my science project.
If the environmentalists could succeed in ridding the atmosphere of CO2, all plant life would die, swiftly followed by all large animal life. It would be left to decay bacteria living off the corpses to replenish the Earth and restart evolution from a low level.
This says Atmospheric carbon has doubled since 1900. As it states Carbon is 0.04% that means in 1900 it was 0.02, which in turn means due to plant life depletion (which would happen if carbon was that low) the carbon reduction would not be reversible. This would mean what carbon we have (or had) would escape the atmosphere and we would all be looking forward to having a planet just like Mars. Given the scaremongering here can’t manage the simple stuff, I wouldn’t trust the rest of it too much.
Forget it Clint, you’ll never convince their closed minds. Its years and years of programming.
As almost 1% of the atmosphere is Argon, I wonder how it is distributed in the atmosphere from the ground to TOA. Is it more abundant, in terms of consentration/ppmv, at the ground than the upper atmosphere? It is a heavy gas that does not mix with other gases, with at molar mass at 40g/mol, more than twice as heavy as air (16g/mol), I would assume it was more abundant at the lower atmosphere, but is this correct?
Under the Obama administration the EPA convinced the Supreme Court that Carbon Dioxide should be considered a pollutant and regulated under the Clean Air Act. That then let the EPA set emission standards for CO2 that eliminated any coal-fired power generation.
Sorry to break it to you but carbon dioxide is not a pollutant! That is shear propaganda.