The History of Oxygen in Earth’s Atmosphere
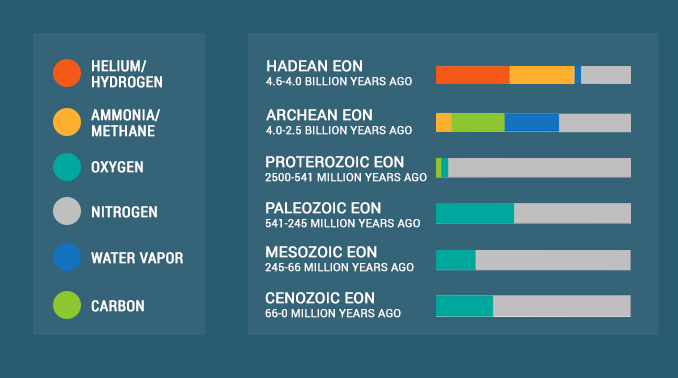
History of Oxygen in Earth’s Atmosphere
Oxygen isn’t the most abundant gas in Earth’s atmosphere composition. Based on the relative volumes of the gases in Earth’s atmosphere, nitrogen is 3 times greater than oxygen.
Because the troposphere is the lowest atmosphere layer, it contains 75 percent of the atmosphere’s mass.
From largest to smallest, Earth’s atmosphere composition contains nitrogen, oxygen, argon, CO2, and trace gases.
But it never used to be like this in the past. Over time, Earth’s oxygen levels have changed significantly with varying levels of hydrogen, helium, carbon dioxide, nitrogen, and oxygen.
1. Hydrogen and helium were in the Hadean Eon
Earth’s early atmosphere was enriched with hydrogen and helium gases. But over time, Earth lost these gases because it wasn’t large enough to hold onto them.
Atmospheric composition is related to escape velocity. The escape velocity of Earth is the speed at which a free object must travel to escape into space from a planet’s gravitational pull.
To this day, Earth loses about 3 kg of hydrogen every second. But Jupiter, Saturn, Uranus, and Neptune hold on tight to these gases because they are much bigger in size. In fact, their atmospheres are mostly these gases.
“Hydrogen and helium gases filled Earth’s early atmosphere. But over time, these gases escaped because Earth wasn’t large enough to hold onto them.”
2. Carbon dioxide in the Archean Eon
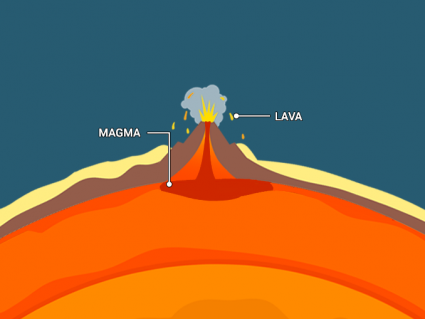
Earth continued to cool and rocks began to solidify in the Archean Eon. Plate tectonics and volcanic activity characterized this early period in Earth’s history.
After hydrogen and helium atoms escaped the atmosphere in the Hadean Eon, the atmosphere mostly consisted of the following gases:
- Methane
- Ammonia
- Water vapor
- Nitrogen
CO2 played a dominant role early in Earth’s history. Earth was uninhabitable for life at this time because the atmosphere was without oxygen.
The only life forms that could exist were anaerobic cyanobacteria (blue-green algae). It turns out that these microorganisms laid the foundation for enriching the atmosphere with oxygen.
3. The Great Oxygenation Event filled the air with oxygen
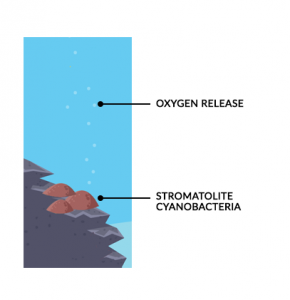
The Great Oxygenation Event marks a time in our geologic history when free oxygen filled the atmosphere. The key to their existence was that they lived without oxygen.
Over time, these cyanobacteria released oxygen as waste. They generated so much oxygen, that it kept building up in the oceans. Eventually, oxygen entered the atmosphere and started an oxygen crisis of its own.
And after oxygen filled the air, it created a habitable planet. Since the early formation of Earth, oxygen levels have changed significantly. For example, free oxygen levels peaked just before the era of dinosaurs.
4. What is Earth’s current atmosphere composition?
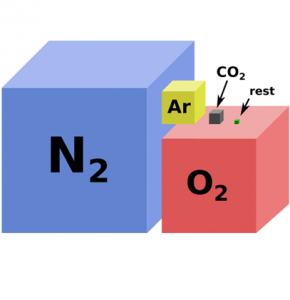
Oxygen plays a pivotal role in aerobic life such as in humans. But oxygen isn’t the most abundant gas in Earth’s atmosphere composition. Based on the relative volumes of the gases in Earth’s atmosphere, nitrogen is actually more than 3 times more abundant than oxygen.
We commonly refer to nitrogen (N2) as an inert gas. But it forms nitrogen sulfide with sulfur, nitrogen dioxide with oxygen, ammonia with hydrogen such as in the nitrogen cycle.
While 78.1% of the atmosphere consists of nitrogen, 20.9% is oxygen. So the bulk of the atmosphere’s composition is these two gases. And since the Paleozoic Era, their relative compositions have see-sawed back and forth ever so slightly.
The next most abundant gas is argon which is an inert gas. Argon doesn’t bond or do much in the atmosphere. This is why the argon cycle doesn’t exist in nature.
History of Oxygen in Earth’s Atmosphere
Oxygen is a gas that is essential for sustaining life on Earth. Without it, there would be no life as we know it.
The history of oxygen in Earth’s atmosphere has been linked to many different events in the past and the level of oxygen was never consistent.
If you have any questions or comments, please use the comment form below to let us know what’s on your mind.









The Earth used to have helium and hydrogen gas. In the back of my mind I almost started suspecting this. Makes sense. Venus holds on to gases too.
I wonder if thicker atmospheres would have bigger fires.
Explain the process of converting nitrogen to helium…
“…nitrogen three times more abundant…”
If I have 80 cents and you have 20 cents wouldn’t most folks say I have 4x as much?
Why no love for water vapor in the graphic? Ten times the volume of CO2. Square cubes for volume are hard to interpret. Perhaps a stack of 2500 colored poker chips better shows the relationships.
I wonder what stabilized oxygen levels in more recent times.
More the toxicity to life, or more fires, or something else …
Btw:
Thanks for using the atmosphere composition info-graphic I made.
It’s CC0 for anyone curious.
I’m greatly interested in actually O2 levels that is thought to have existed. What was the highest concentration, when and how caused the gyrations mentioned.
Thank you for your informative educational website.